Don't Worry About Those Safety Signals...The "Experts" Say They Don't Matter
What's A Heart Attack Here And There Among Friends?
The corporate media wants you to overlook the fact that the CDC lied about the safety data on the COVID-19 mRNA inoculations accumulated in the Vaccine Adverse Event Reporting System (VAERS).
On the one hand, we have the CDC, in its Morbidity and Mortality Weekly Report1, touting the benefits of the latest “bivalent” booster mRNA injections from Pfizer.
Bivalent booster doses provided additional protection against COVID-19–associated emergency department/urgent care encounters and hospitalizations in persons who previously received 2, 3, or 4 monovalent vaccine doses.
Yet not only does the data argue strongly against the CDC’s narrative position, but peer-reviewed articles in leading medical journals refute part or all of the CDC’s assessment of the bivalent inoculations. In the case of the mRNA inoculation refutations, the articles and op-eds opposing the booster shots are backed up with clinical research data.
The utility of the booster shots is seriously questioned by Dr. Paul Offit2, writing in a recent issue of the New England Journal of Medicine, pointing out that the bivalent booster targeted strains no longer dominant in the wild.
As of November 15, 2022, only about 10% of the population for whom the bivalent vaccine had been recommended had received it.5 By December 2022, the BA.4 strain was no longer circulating, and BA.5 accounted for less than 25% of circulating SARS-CoV-2 strains, having been partially replaced by more immune-evasive strains, such as BQ.1, BQ.1.1, BF.7, XBB, and XBB.1.
This stance was and is supported by the CDC’s own variant monitoring which shows both BA.5 and BA.4.4 declining in prevalence throughout the fall of 2022.
With only 10% of the population having received the bivalent booster by November 15, it is simply not rational to argue the booster played any role in reducing and eliminating either Omicron subvariant. Even if a role could be interpolated for the booster in eliminating either subvariant, that fails to explain how other subvariants (such as XBB.1.5) filled the void—if the inoculation-generated antibodies actually protected against XBB.1.5 and other variants, there would be no successor strain to displace BA.4 or BA.5.
Offitt supports his contention with two studies released on the BioRxiv Preprint Server in October.
The first one3 found the boosters did not do a superior job of generating antibodies.
When given as a fourth dose, a bivalent mRNA vaccine targeting Omicron BA.4/BA.5 and an ancestral SARS-CoV-2 strain did not induce superior neutralizing antibody responses in humans, at the time period tested, compared to the original monovalent vaccine formulation.
Update: Wang’s research was published in NEJM4 on January 11, 2023, and is officially peer-reviewed material. Offit's own NEJM article cites the preprint version.
The second study5 suggested that “immune imprinting” inhibited the booster’s efficacy.
Our findings suggest that immune imprinting by prior antigenic exposure may pose a greater challenge than currently appreciated for inducing robust immunity to SARS-CoV-2 variants
Functionally, the bivalent approach appears to be less efficacious than a targeted monovalent approach might be, according to Offit.
Why did the strategy for significantly increasing BA.4 and BA.5 neutralizing antibodies using a bivalent vaccine fail? The most likely explanation is imprinting. The immune systems of people immunized with the bivalent vaccine, all of whom had previously been vaccinated, were primed to respond to the ancestral strain of SARS-CoV-2. They therefore probably responded to epitopes shared by BA.4 and BA.5 and the ancestral strain, rather than to new epitopes on BA.4 and BA.5. This effect could possibly be moderated by immunizing people either with BA.4 and BA.5 mRNA alone or with a greater quantity of BA.4 and BA.5 mRNA. Evidence in support of these strategies can be found in Pfizer–BioNTech’s data regarding its BA.1-containing bivalent vaccine, which showed that BA.1-specific neutralizing-antibody responses were greater in persons who were injected with a monovalent vaccine containing 30 μg or 60 μg of BA.1 mRNA or a bivalent vaccine containing 30 μg of BA.1 mRNA and 30 μg of ancestral-strain mRNA than in those who received a bivalent vaccine containing 15 μg of each type of mRNA.
Offit’s NEJM analysis was noted by several corporate media outlets, including the Huffington Post, NBC News, and Time Magazine
Shortly after Offit’s NEJM article, in the wake of Lisa Marie Presley’s untimely death from cardiac arrest, a brief article in Fortune Magazine threw more than a little shade on those who speculated about whether the mRNA inoculations might have contributed to her death.
Shortly after news broke that 54-year-old singer Lisa Marie Presley had died of cardiac arrest, some people were once again pointing blame at the COVID-19 vaccine. Speculation regarding a connection between the COVID-19 vaccine and cardiac arrest resurfaced after 24-year-old Buffalo Bills safety Damar Hamlin was rushed off the field during a Monday Night Football game earlier this month after suffering cardiac arrest. But experts maintain there is no link between the two.
“The [suggested] link between the COVID-19 vaccine [and cardiac arrest] is wildly and irresponsibly speculative from a very vocal minority,” Dr. Michael Emery, cardiologist and co-director of the sports cardiology center at Cleveland Clinic, previously told Fortune.
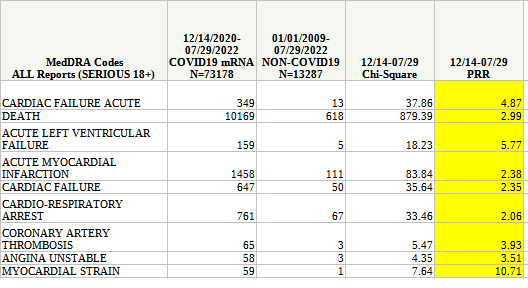
However, the CDC’s own analysis of the VAERS database shows that, for just about all types of major cardic events, there are strong safety signals that practically demand urgent investigation.
The CDC, of course, is doing little or nothing to investigate these safety signals.
Yet, given that so many forms of cardiac distress show strong safety signals regarding the COVID mRNA inoculations, speculation about whether the shots caused either Lisa Marie Presley’s death is not only reasonable, it arguably becomes imperative—how else will the CDC be pressured to do the job it should have done all along, which is to monitor the administration of the inoculations for precisely these safety signals.
Moreover, the same day the article about Lisa Marie Presley’s passing appeared in Fortune, the CDC announced a possible “safety signal” correlating the mRNA booster shots with ischemic stroke in people 65 or older.
Following the availability and use of the updated (bivalent) COVID-19 vaccines, CDC’s Vaccine Safety Datalink (VSD), a near real-time surveillance system, met the statistical criteria to prompt additional investigation into whether there was a safety concern for ischemic stroke in people ages 65 and older who received the Pfizer-BioNTech COVID-19 Vaccine, Bivalent. Rapid-response investigation of the signal in the VSD raised a question of whether people 65 and older who have received the Pfizer-BioNTech COVID-19 Vaccine, Bivalent were more likely to have an ischemic stroke in the 21 days following vaccination compared with days 22-42 following vaccination.
As might be expected, both Pfizer and Moderna rejected even the possibility.
Pfizer and BioNTech said in a statement that they have been made aware of limited reports of ischemic strokes in people 65 and older following vaccination with their updated shot.
"Neither Pfizer and BioNTech nor the CDC or FDA have observed similar findings across numerous other monitoring systems in the U.S. and globally and there is no evidence to conclude that ischemic stroke is associated with the use of the companies' COVID-19 vaccines," the companies added.
For its part, the CDC promptly discounted the safety issue, more or less adopting the position of Pfizer and Moderna.
The Centers for Disease Control and Prevention on Friday said it is “very unlikely” the Pfizer omicron booster carries a risk of stroke for seniors after it launched an investigation into a preliminary safety concern detected by one of its monitoring systems.
However, we should keep in mind that the CDC’s own analysis of the VAERS data concluded there was a strong safety signal.
The potential for the mRNA inoculations to cause a variety of stroke events is, according to the CDC, quite significant—yet we are to believe such events are “very unlikely”.
The nature of the PRR analysis is to establish what safety signals are, in fact, “likely”, and any safety signal with a PRR value greater than two is a safety signal that is, by definition and by varying degrees, quite “likely”. The PRR values above 2 mean that what was once a mere possibility must now be upgraded to a probability.
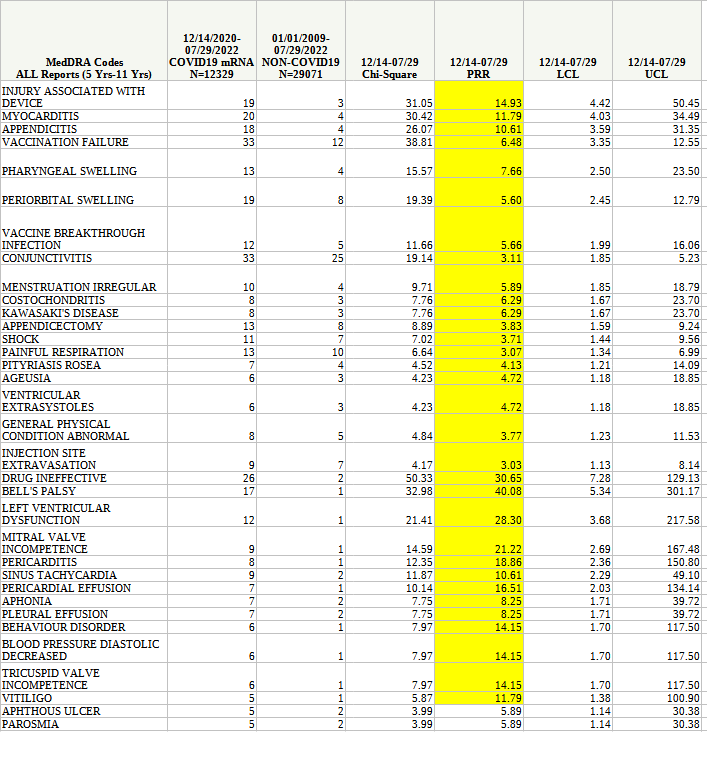
Separately, the CDC has sought to assure the public that side effects from the bivalent booster shots among the 5-11 patient demographic were “rare”.
“Preliminary safety findings from the first 11 weeks of bivalent booster vaccination among children aged 5-11 years are reassuring,” the study said.
These are but a few of the safety signals correlated to the mRNA booster shots administered to children ages 5-11.
That doesn’t look “rare”.
Adverse events may not be happening with every other injection, but they most definitely are happening, and they most definitely impact everyone’s ongoing assessment of personal risk when deciding whether to get the shots or have them administered to one’s children.
This dogged insistence by the legacy media to ignore indisputable facts and evidence regarding COVID-19 and the mRNA inoculations is why those facts must be published, republished, and then published again. Because the media wants to pretend this evidence does not exist, the imperative is to remind one and all that the evidence very much does exist.
Some of the evidence extends back to the earliest days after the mRNA inoculations were released and inflicted upon a credulous and unsuspecting public. Proofs of the CDC’s and FDA’s mendacity, corruption, and incompetence abound, yet the corporate media will not even acknowledge these proofs most of the time.
Yet the data does exist. The proofs do exist. The evidence most assuredly is out there.
People deserve to have that data, so they can make their own assessments about the utility and safety of the mRNA inoculations. Without all the data, without all the facts, there can be no such thing as “informed consent”—and that alone makes the fullest publication of all the facts, evidence, and data regarding the mRNA inoculations a moral imperative.
All facts really do matter, especially where matters of personal health and well being are concerned.
Tenforde, M. W., et al. “Early Estimates of Bivalent MRNA Vaccine Effectiveness in Preventing COVID-19–Associated Emergency Department or Urgent Care Encounters and Hospitalizations Among Immunocompetent Adults — VISION Network, Nine States, September–November 2022.” Morbidity and Mortality Weekly Report, vol. 71, no. 51–52, 2022, doi: http://dx.doi.org/10.15585/mmwr.mm715152e1.
Offit, P. A. “Bivalent Covid-19 Vaccines — A Cautionary Tale.” New England Journal Of Medicine, 2023, doi: 10.1056/NEJMp2215780.
Wang, Q., et al. Antibody Responses to Omicron BA.4/BA.5 Bivalent MRNA Vaccine Booster Shot. 24 Oct. 2022, https://www.biorxiv.org/content/10.1101/2022.10.22.513349v1.
Wang, Q., et al. “Antibody Response to Omicron BA.4–BA.5 Bivalent Booster.” New England Journal of Medicine, 2023, doi: 10.1056/NEJMc2213907.
Collier, A. Y., et al. Immunogenicity of the BA.5 Bivalent MRNA Vaccine Boosters. 25 Oct. 2022, https://www.biorxiv.org/content/10.1101/2022.10.24.513619v1.






They're in too deep.