COVID-19: The Endless "Emergency"
The "Enabling Act" For Pharmaceutical Authoritarianism
In what has become an almost formulaic 90-day ritual, Health and Human Services Secretary “consulted”….someone (the voices in his head, perhaps?), scratched, thought, and then realized that COVID-19 is still a serious threat to the public health and that, by gosh, he’s got to do something!
By “something” I mean he renewed the emergency declaration issued by his predecessor all the way back in January of 2020.
US Department of Health and Human Services Secretary Xavier Becerra officially renewed the declaration, extending it through October 13, 2022.
The emergency declaration has been in place since January 2020, and the latest renewal comes as the Omicron offshoot BA.5, the most contagious variant yet, continues to stake its claim in the US. Daily case rates, though vastly undercounted, are the highest they've been in months, as are Covid-19 hospitalizations and deaths.
The pandemic will last for another 90 days at least. Unless Becerra decides to extend the pandemic beyond that. Which he may, given hints in April that the extension issued then might be the last one.
It remains unclear whether HHS will extend the emergency for a second time this summer, but providers will likely know the answer in a month. HHS Secretary Xavier Becerra has promised a 60-day heads-up to providers and states that the PHE will end.
Despite the speculation in the media of an end to the public health emergency declaration, Secretary Becerra went ahead with the extension until October—at which point he could easily extend it again for another three months, and another, and another, for as long as he is HHS Secretary.
What Makes COVID-19 A “Public Health Emergency”?
What are the criteria for declaring a public health emergency?
Simply put, there are none.
There are no scientific criteria. There are no objectively verifiable metrics which are applied. There are no data sets that must be accumulated. A public health emergency exists whenever the Secretary of Health and Human Services says a public health emergency exists.
That is how his authority to declare an emergency reads in Section 319 of the Public Health Service Act, codified in the US Code under 42 USC 247d.
(a) Emergencies
If the Secretary determines, after consultation with such public health officials as may be necessary, that—
(1) a disease or disorder presents a public health emergency; or
(2) a public health emergency, including significant outbreaks of infectious diseases or bioterrorist attacks, otherwise exists,
the Secretary may take such action as may be appropriate to respond to the public health emergency, including making grants, providing awards for expenses, and entering into contracts and conducting and supporting investigations into the cause, treatment, or prevention of a disease or disorder as described in paragraphs (1) and (2). Any such determination of a public health emergency terminates upon the Secretary declaring that the emergency no longer exists, or upon the expiration of the 90-day period beginning on the date on which the determination is made by the Secretary, whichever occurs first. Determinations that terminate under the preceding sentence may be renewed by the Secretary (on the basis of the same or additional facts), and the preceding sentence applies to each such renewal. Not later than 48 hours after making a determination under this subsection of a public health emergency (including a renewal), the Secretary shall submit to the Congress written notification of the determination.
The wording of the statute places almost no restrictions on the HHS Secretary’s power to declare a public health emergency. HHS can declare flatulence to be a public health emergency, or even rap music.
A public health emergency declaration is a purely arbitrary, even capricious power given to the Executive Branch, with no apparent statutory limits to be respected.
Does COVID-19 Really Look Like A Plausible “Emergency”?
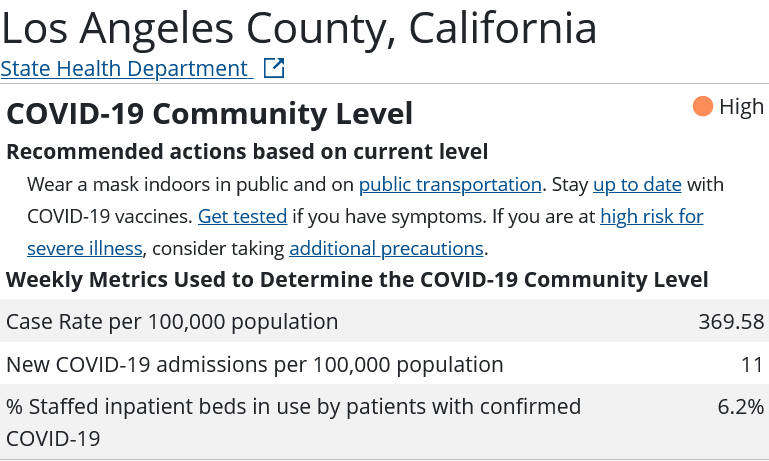
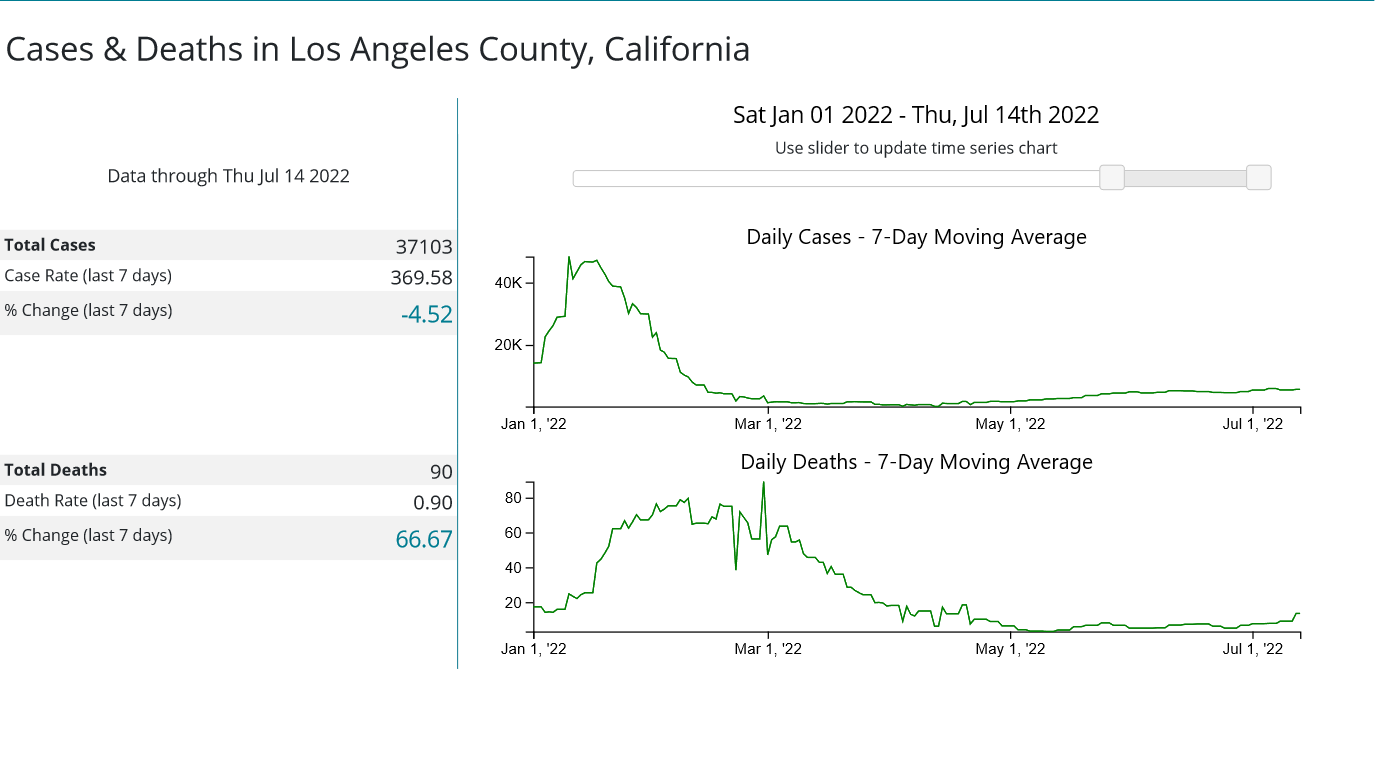
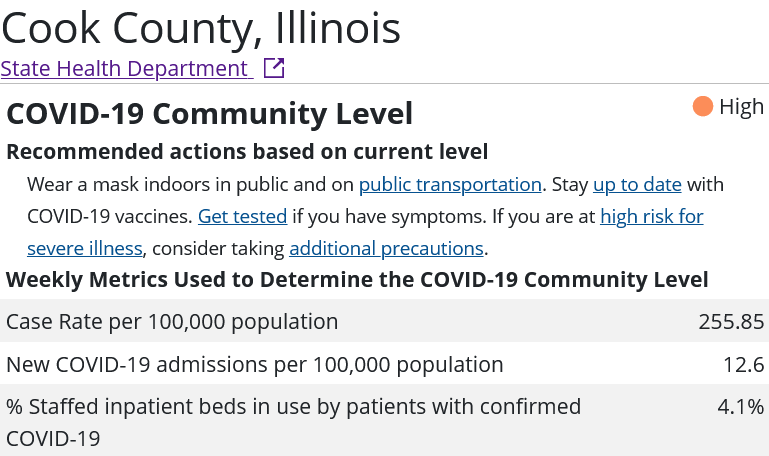
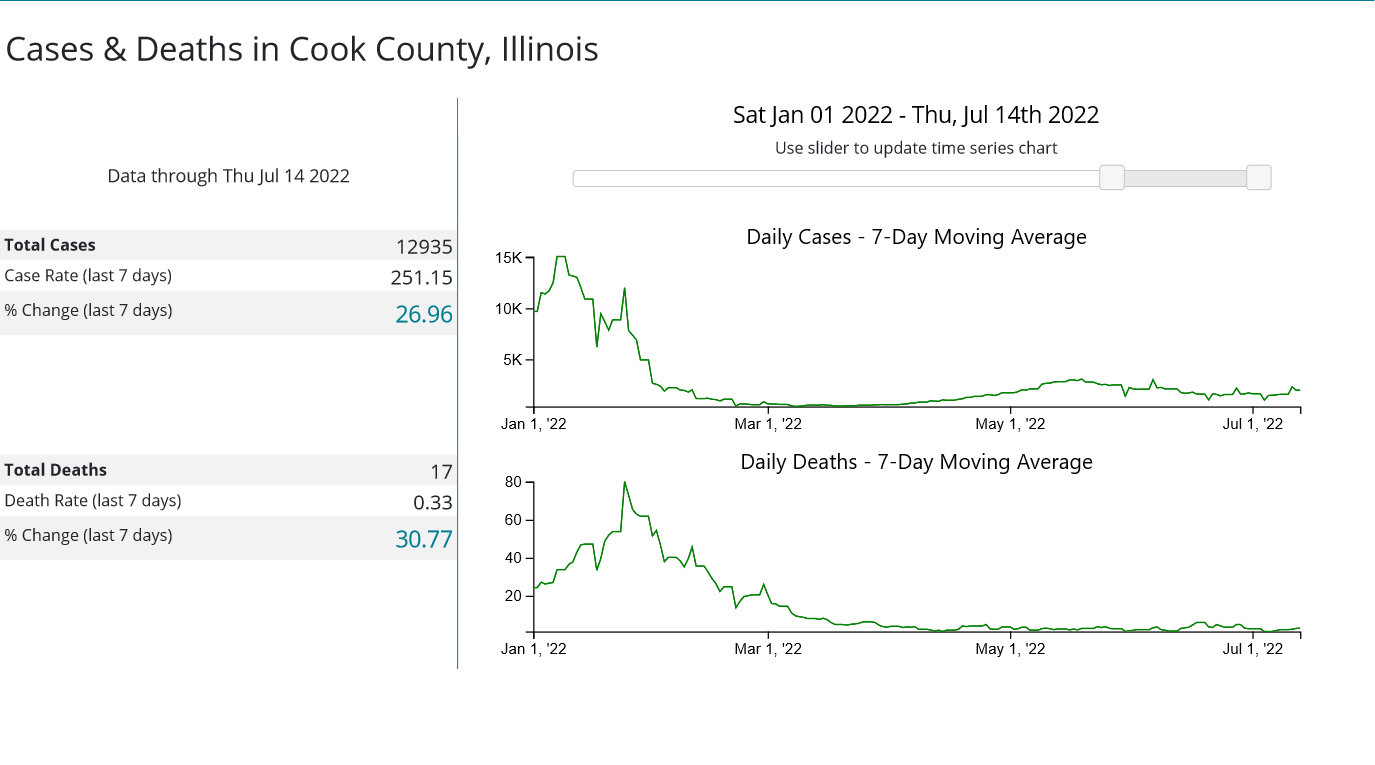
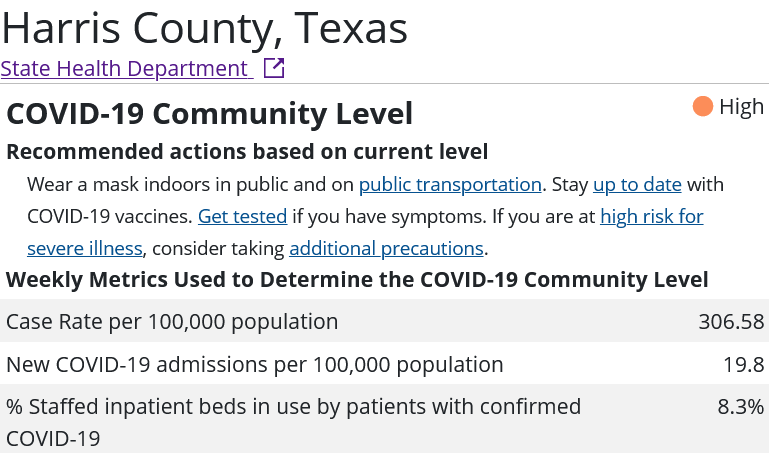
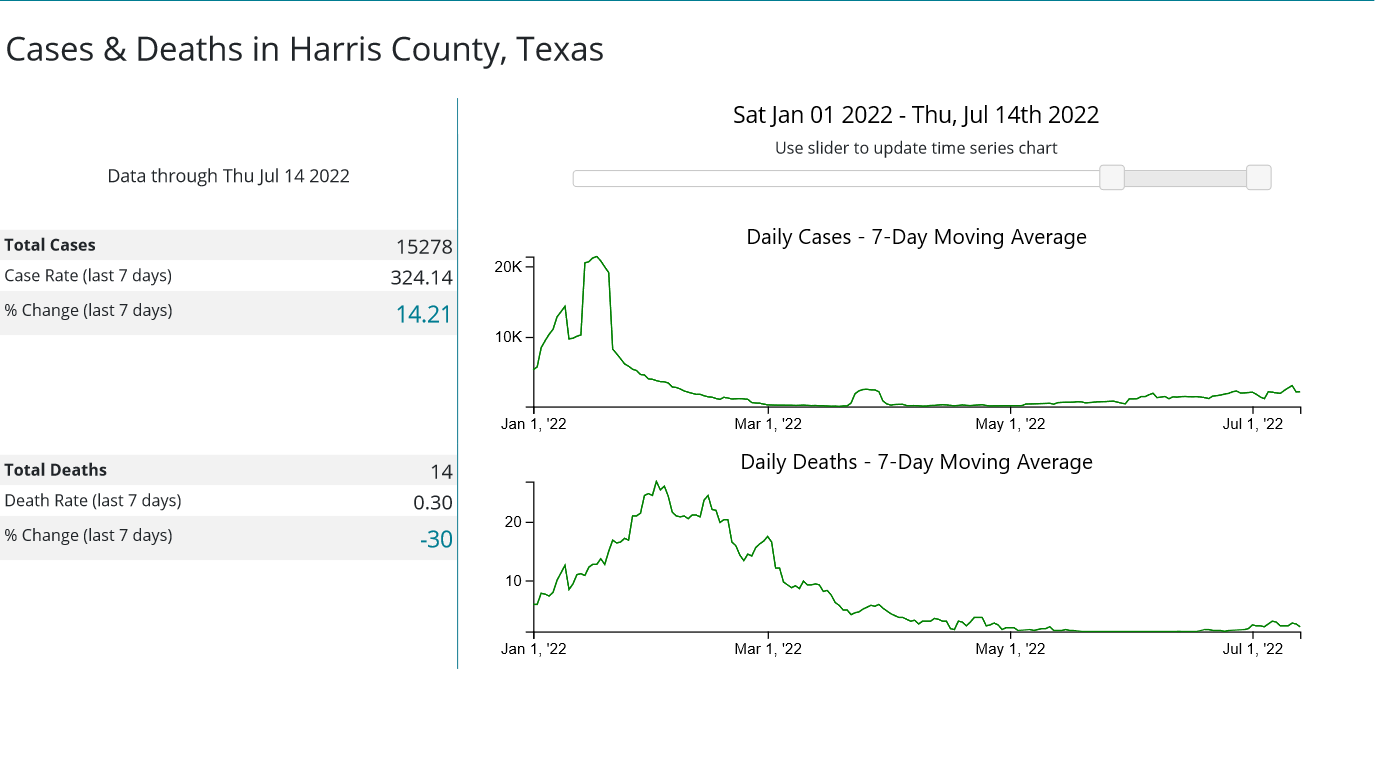
Is Secretary Becerra being arbitrary and capricious with his renewal of the declaration? Consider the statistics assembled by the CDC for the three most populous counties in the United States: Los Angeles County, California; Cook County, Illinois; and Harris County, Texas.
Whether or not any of these counties appear to be in the midst of a COVID-19 emergency I leave to the reader to decide. However, if these counties are not in the midst of a COVID-19 emergency, then Secretary Becerra is being arbitrary and capricious in his exercise of his Section 319 authority. It is as simple as that.
What Changes In An Emergency?
The primary impact of an emergency declaration is the ability to tap into funds set aside within the HHS budget specifically for emergency spending. An emergency declaration also gives the Secretary the authority to move funds and personnel around within the HHS bureaucracy as needed.
Perhaps the most impactful consequence of a Section 319 emergency declaration that it opens the door to FDA Emergency Use Authorizations of various unapproved drugs and medical devices, under section 564 of the Federal Food, Drug, and Cosmetic act, codified in the US Code as 21 USC 360bbb-3.
(b) Declaration of emergency or threat justifying emergency authorized use
(1) In general
The Secretary may make a declaration that the circumstances exist justifying the authorization under this subsection for a product on the basis of—
(C) a determination by the Secretary that there is a public health emergency, or a significant potential for a public health emergency, that affects, or has a significant potential to affect, national security or the health and security of United States citizens living abroad, and that involves a biological, chemical, radiological, or nuclear agent or agents, or a disease or condition that may be attributable to such agent or agents;
In short, before the FDA can grant Emergency Use Authorization (EUA) to Pfizer for their (toxic and useless) mRNA inoculation against the SARS-CoV-2 virus, there has to first be a public health emergency. An EUA itself does not extend past the expiration of the public health emergency declaration.
Note that all of the COVID-19 immunization products, including Novovax, the Janssen adenovirus vector inoculation, as well as the Pfizer-BioNTech and Moderna mRNA inoculations, are made available in the US under the aegis of an Emergency Use Authorization. Additionally, COVID-19 therapeutics Paxlovid and Lageviro (molnupiravir) are also made available under Emergency Use Authorization. None of these products have been approved under normal FDA approval regulations.1
Section 319 An “Enabling Act” For Bypassing FDA Approval Process
As a direct consequence of the repeated extensions of the original 2020 public health emergency declaration, the HHS Secretary’s authority to declare a public health emergency under Section 319 of the Public Health Service Act has become, disturbingly, an “Enabling Act” for bypassing the normal FDA approval process for new vaccines and drugs.
Under the rubric of “Emergency Approval”, the FDA has seen fit to authorize new inoculation preparations with little or no clinical trial data to support either efficacy or safety. As Toby Rogers has already outlined on his Substack “UTobian”, the FDA is already doing this, casting scientific rigor to the winds in favor of arbitrary and capricious exercise of bureaucratic authority.
Thus Section 319 of the Public Health Service Act becomes the Enabling Act for pharmaceutical authoritarianism.
Big Pharma’s reliance on the EUA process rather than the regular approval process carries the consequence that, should the public health emergency declaration expire—which it automatically does every 90 days without renewal—all COVID-19 vaccines, inoculations, therapeutics, tests, devices, and anything else devised to combat COVID-19 and made available under EUA, would immediately be rendered unavailable.
If/when the public health emergency lapses, Pfizer must either produce and distribute Comirnaty properly or watch its mRNA inoculation get pulled from the shelf.
If/when the public health emergency lapses, Pfizer’s EUA for Paxlovid lapses. Merck’s EUA for molnupiravir lapses. The EUA for Novavax lapses.
Is Becerra extending the emergency declaration ad infinitum simply to gratify Big Pharma’s lust for COVID-19 profits? There is, so far as I am aware, no publicly available data that would prove such an allegation. While an absence of proof is never proof of absence, to ascribe such a mercenary motive to Becerra’s emergency extensions without proof is merely speculation.
What is not speculation is the CDC county-by-county metrics on COVID-19 around the country. Do those metrics support Secretary Becerra’s Public Health Emergency declaration?
You tell me.
While Pfizer’s mRNA inoculation (jointly developed with BioNTech) did receive full FDA approval under the brand name Comirnaty, in issuing its approval the FDA also maintained separately the EUA for the original Pfizer-BioNtech preparation. Additionally, in its Letters of Authorization for both Comirnaty and the Pfizer-BioNTech preparation, the FDA explicitly stated that the two products are “legally distinct”. A Florida Federal court has held that the FDA’s distinction is substantive and that Comirnaty and the Pfizer-BioNTech inoculations are cannot be substituted the one for the other.






I read an article on one of the news sites yesterday about one of the reasons they can't end it. The emergency lowered, or removed, restrictions on food stamps and medicaid (and probably other programs) They estimated there's something like 20 or 25% extra in those programs right now that wouldn't qualify.
It also eliminated the rule that people had to work.
Ending the emergency would kick a lot of people off the programs during high inflation/recession.
So thinking politically. Extend it to October when a "wave" hits and then it's justified to extend it to Jan when a red wave takes over. Then end it and toss the hand grenade to the Republicans.
No. It's not justified.
Also.
I notice Canadian officials use the term 'at least' too.
I'm convinced the two countries are coordinating their messaging to ram through a medical tyranny.